 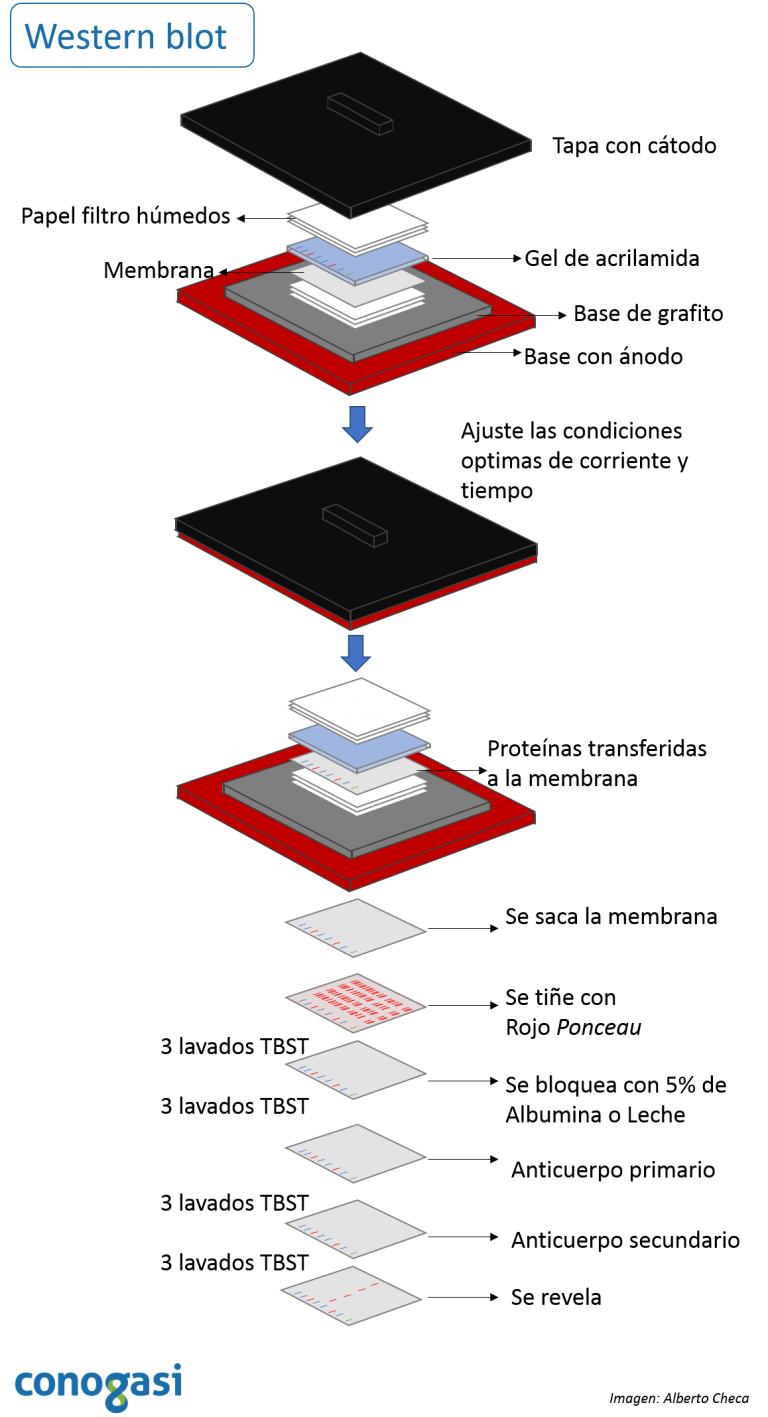
28) Slide the film through the developer to develop. 27) Burn the film for 1min, bend the top right hand corner down and back again to distinguish the correct orientation. When in the darkroom, cut a piece of film to fit the cassette. 26) Bring the cassette, film and scissors in a darkroom with a developer. Wrap the membrane up in the saranwrap and place it into the film cassette right-side up. 25) Drip off excess mix, and transfer the membrane to a clean piece of saranwrap upside down. 24) Use tweezers to move membrane on to a piece of saranwrap and drip the reagent mix all over the membrane. 23) Mix 2ml of 1 and 2ml of 2 in a 15ml tube (BE SURE TO CHANGE PIPETS IN BETWEEN REAGENTS). Developing the blot: 22) Materials needed: Two pieces of saran-wrap, detection reagent 1 and 2 (bottles at 4 C with a big ECL on the label, one with a white top and one with a black top), film cassette, scissors, tweezers and film. 20) Dump off third wash and put in secondary antibody mix. 19) Dilute out secondary antibody 1: 5,000 (1µl in 5ml). 17) Incubate primary antibody overnight at 4 C. Binding the antibody 16) Add diluted primary antibody. 15) Transfer to the smallest container possible. Cut the top right-handcorner so that "up" is easily recognizable. Use a pencil to mark the ladder, and distinguish the 50kDa marker somehow. 13) Rinse with water until markings appear. Leave on the orbital shaker or rocker for about 15 mins. 12) Use tweezers to put membrane into a small tupperware top and cover with ponso (red stuff). Staining the blot: 11) Dump out the TB, and remove the sandwich. 10) Run the blot on manual and constant V at 100V for about 1 hour. Then fill the gel box up to the top of the red half of the sleeve with TB. 9) Put the blotting sandwich into the black and red sleeve with the black part of the sandwich facing the black part of the sleeve. Then put the ice pack in next to the sleeve. 8) Put a small stir bar in the gel box underneath the black and red sleeve. Leave soaked pads in buffer until ready to assemble the blotting sandwich. 7) Roll a 15ml tube up and down and side to side over the completed sandwich to remove any bubbles. Soak blotting pads in transfer buffer, pressing down to remove trapped air bubbles. 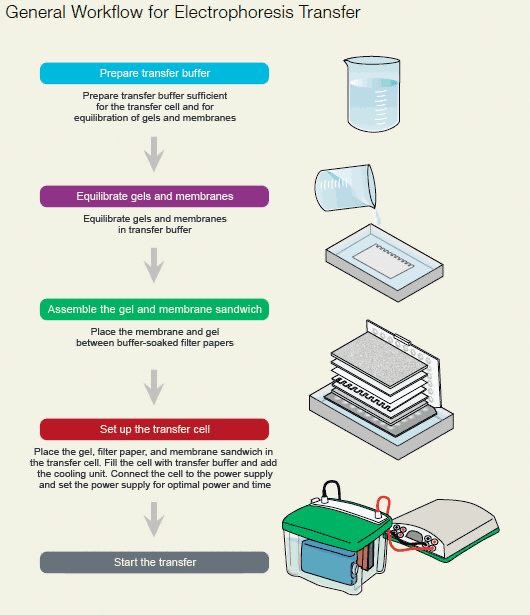
6) To complete the sandwich, one more piece of paper goes on and then the last pad, again making sure both are thoroughly soaked in TB. When it is completely soaked with TB, move it onto the sandwich next. 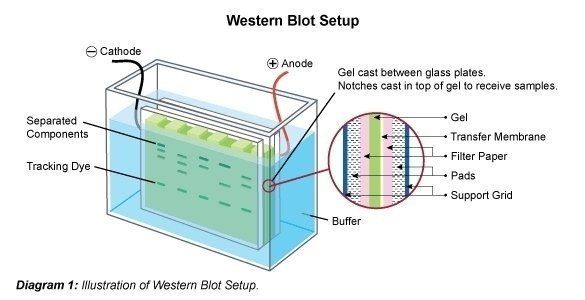
5) Use the tweezers to grab the nitrocellulose membrane and dunk it in the dH2O container, make sure the entire membrane is wet, and then move it to the TB. Then put on a pad and one piece of paper (make sure both are completely wet in the TB). 4) To make the blotting sandwich, begin by putting the blotting apparatus in the TB container with the black side down. To remove the other plate, place the gel in the TB container and gently scrap off with a flat scoopula. 3) Remove one glass plate from the gel by twisting a spacer between the plates. Set up blot: 2) Prepare two tupperware containers, one with dH2O and one with 1/2X Transfer Buffer (TB). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
